Hib Halverson's Big Block From Hell Series
Hib Halverson's Big Block From Hell Series
Stoy-kee-ah...I'm sorry, what?
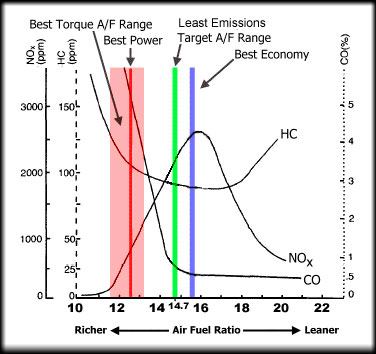
This chart shows the air-fuel ratio ranges for various engine performance regimes. Pink is the range of AFRs for best torque output. Red is the range of AFRs for maximum power. Green the the range for a combination of low exhaust emissions and good fuel economy and blue is for best gas mileage.
Image: Toyota Motor Sales USA.
The term "stoichiometric" (say "Stoy-kee-ah-metric") is a word chemists and engineers use to describe the air-fuel ratio necessary for complete or "perfect" combustion to occur.
For gasoline, a stoichiometric mixture is about 14.7:1. It may differ a little bit either way depending on how the gasoline is blended and whether or not it has ethanol in it. In theory, stoichiometric combustion has all the hydrocarbons in the fuel oxidized with only heat, water vapor, nitrogen and a very small amount of inert gases going out the exhaust.
In practice, stoichiometric combustion never occurs in a gasoline engine. Modern engines come close but there's always a small amount of unburned hydrocarbons and carbon monoxide which occur do to the quenching effects of the combustion chamber walls and piston surfaces along with the limited time available for combustion-a matter of a few milliseconds when the engine is at high rpm.
Attempting to get the part throttle air-fuel ratio near stoichiometric is an admirable goal because stoichiometric combustion produces the least amount of exhaust emissions and, because of its efficiency, results in good fuel economy.
Is a stoichiometric AFR what gets you the best gas mileage? Actually, not. The AFR for best fuel economy is about 15.5:1 and, again, that may differ a little depending on the gasoline blend and how much ethanol is in it. Modern engines with computer controls and catalytic converters don't run that lean because AFRs more lean than stoichiometric can damage cats. With engines in pre-1975 cars, like my old hot rod, cat life is not an issue, so AFR when the car is cruising at part-throttle, can be that lean for better gas mileage.
But...there's a catch. To run the engine between 14.7 and 15.5 AFR at part-throttle with no lean misfire requires robust ignition hardware. Engines with stock-based ignition systems which ran fine when the part-throttle AFR was 13.5:1 or so, may not have the "stuff" to fire a mixture that is running at 14.5-15.5:1.
Additionally, engines set-up to run at 14.5:1-15.5:1 at part throttle must have spark advance curves which are properly calibrated. Excessive spark advance at part-perhaps due to too much vacuum advance or overly-aggressive centrifugal advance curve-will cause detonation. A good starting point for vacuum advance with an engine having a Holley Double Pumper calibrated for good street drivability is 8-10°. Often, that's less than what a stock distributor's vacuum advance "can" will provide and the vacuum advance will have to be limited. As for the centrifugal advance curve, watch out for too much advance at low to mid-range engine speeds.
Lastly, once load on the engine begins to build, you need to make sure the carburetor's power enrichment functions are enabled at the right point to prevent detonation under medium-load conditions. With our engine it was 7.5-in. of vacuum but, depending on the engine configuration, gearing and vehicle weight, it may need to be anywhere from 5.5-in. to 10.5-in. vacuum.